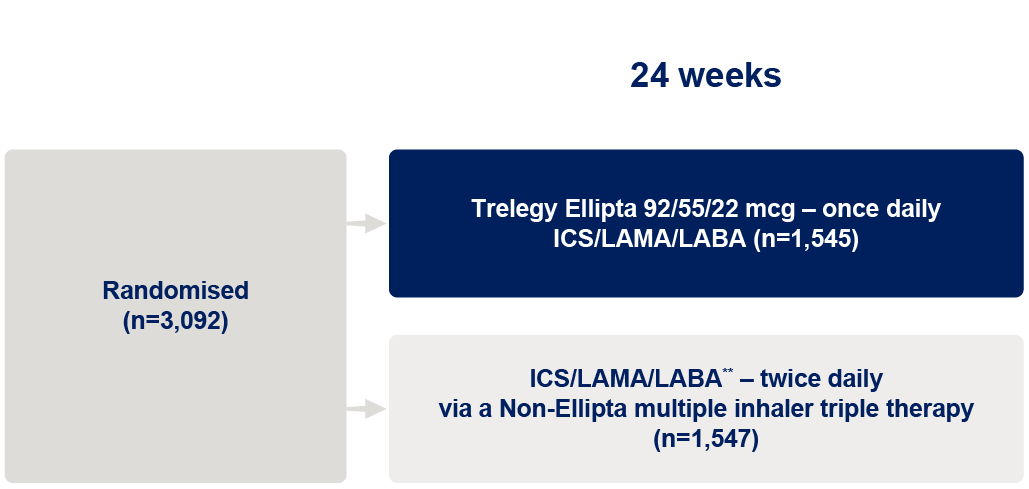
Study Design
A real world, phase IV, randomised, open-label, multicentre, effectiveness study set in usual clinical practice. Trial conducted across 147 centres in the UK, Germany, the Netherlands, Spain and Sweden.
Endpoints
Primary endpoint:
- Proportion of CAT responders* at Week 24.
Secondary endpoints included:
- Change from baseline in FEV1 at 24 weeks*
- Percentage of participants making at least one critical error in inhalation technique at 24 weeks ++
- Treatment-related AEs
++ In a subset of participants. Critical errors were defined as an error that is likely to result in no, minimal or significantly reduced medication being inhaled.
Population
- ≥40 years of age
- Symptomatic COPD (CAT score ≥10 at screening)
- Receiving Non-Ellipta maintenance therapy for ≥16 weeks prior to randomisation (Multiple Inhaler Triple Therapy; ICS+LABA; LAMA+LABA)
- History of ≥1 COPD moderate/severe exacerbation* in the 3 years prior to randomisation
Results
CAT score responders (primary endpoint)
Trelegy Ellipta showed a significant improvement in CAT score responders* vs. multiple inhaler triple therapy at Week 24 (47% vs. 40% respecively. OR 1.31, p<0.001)< /p>
*Response is defined as a reduction from baseline of CAT score = or >2 at 24 weeks
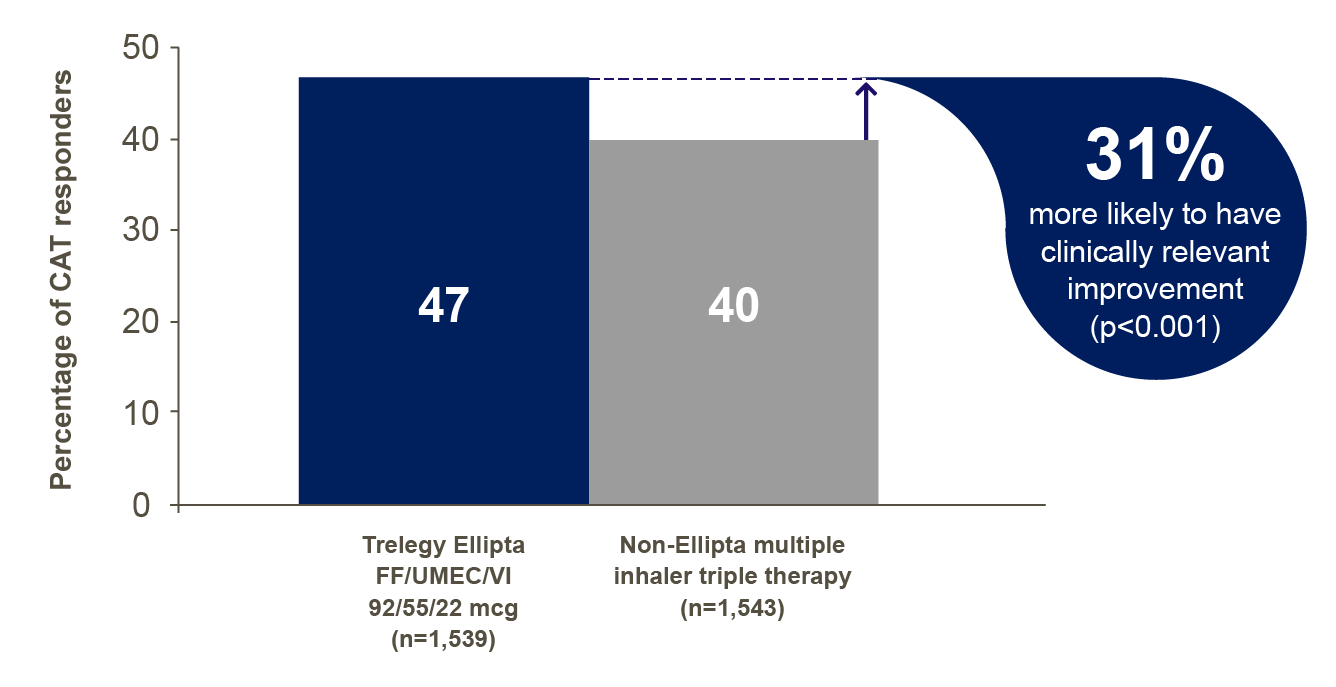 Trelegy Ellipta patients were 31% more likely than patients on multiple inhaler triple therapy to have a clinically relevant improvement (OR 1.31 [95% CI 1.13,1.51]:p<0.001). Patients who modified their
randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy were considered as non-responders. CAT data for patients who discontinued randomised treatment without receiving
another COPD maintenance treatment therapy during the study was included.
Trelegy Ellipta patients were 31% more likely than patients on multiple inhaler triple therapy to have a clinically relevant improvement (OR 1.31 [95% CI 1.13,1.51]:p<0.001). Patients who modified their
randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy were considered as non-responders. CAT data for patients who discontinued randomised treatment without receiving
another COPD maintenance treatment therapy during the study was included.
Lung function (secondary endpoint)
Trelegy Ellipta significantly improved lung function vs. non-Ellipta multiple inhaler triple therapy
 Patients who modified their randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy were considered as non-responders. CAT data for patients who
discontinued randomised treatment without receiving another COPD maintenance treatment therapy during the study was included. Week 24 data was used regardless of whether patients
discontinued/modified their randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy.
Patients who modified their randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy were considered as non-responders. CAT data for patients who
discontinued randomised treatment without receiving another COPD maintenance treatment therapy during the study was included. Week 24 data was used regardless of whether patients
discontinued/modified their randomised treatment, changed pulmonary rehabilitation status or started oxygen therapy.
Safety

Pneumonia rates were similar between Trelegy and non-ellipta MITT

